
In this paper, we have adapted the clustered regularly interspersed palindromic repeats (CRISPR) system from Streptococcus pyogenes to repress genes in trans in the cyanobacterium Synechococcus sp. Trans-acting regulators provide novel opportunities to study essential genes and regulate metabolic pathways. Our combinatorial method using a Cas9 nickase genome editing tool to integrate the gene repression module demonstrates easy-to-use and high-efficiency advantages, paving the way for stably manipulating genes, even essential ones, for functional characterization and microbial engineering. These results indicate the effectiveness of tandem promoter-driven RNA repression modules in promoting gene repression in chromosomal integrants. With the design of a tandem repetitive promoter-driven asRNA module and the use of a new Cas9 nickase genome editing tool, a chromosomal integrant (LM3P) was generated in a single step and successfully enhanced RNA repression, with a 27% decrease in acetate titer on cellulose in antibioticfree medium. However, further testing on chromosomal integrants that were created by double-crossover recombination exhibited only very weak repression because DNA integration dramatically lessened gene dosage. The strains with pta gene repression generally diverted more carbon into ethanol. To control both acetate and more » lactate simultaneously, we transformed the repression plasmid into lactate production-deficient mutant and found the plasmid delivery reduced acetate titer by more than 33%, concomitant with negligible lactate formation. Our plasmid-based expression of antisense RNA (asRNA) molecules targeting the phosphotransacetylase (pta) gene successfully reduced the enzymatic activity by 35% in cellobiose-grown cells, metabolically decreased the acetate titer by 15 and 52% in wildtype transformants on cellulose and xylan, respectively. Here we developed a Cas9 nickase-assisted chromosome-based RNA repression to stably manipulate essential genes in C. Clostridium cellulolyticum is a promising candidate for consolidated bioprocessing however, its genetic manipulation to reduce the formation of less-valuable acetate is technically challenging due to the essentiality of acetate-producing genes. ELSAs enable simultaneous and stable regulation of many genes for metabolic engineering and synthetic biology = ,Įssential gene functions remain largely underexplored in bacteria. Using ELSAs, we created three highly selective phenotypes in Escherichia coli, including redirecting metabolism to increase succinic acid production by 150-fold, knocking down amino acid biosynthesis to create a multi-auxotrophic strain, and repressing stress responses to reduce persister cell formation by 21-fold. 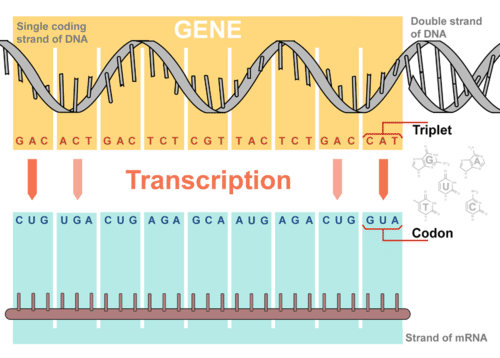 
We design ELSAs by combining non-repetitive genetic parts according to algorithmic rules quantifying DNA synthesis complexity, sgRNA expression, sgRNA targeting, and genetic stability. We apply biophysical modeling, biochemical characterization, and machine learning to develop toolboxes of non-repetitive genetic parts, including 28 sgRNA handles that bind Cas9. We stably co-expressed 22 sgRNAs within non-repetitive extra-long sgRNA arrays (ELSAs) to simultaneously repress up to 13 genes by up to 3500-fold. When using CRISPR interference, co-expressing many single-guide RNAs (sgRNAs) triggers genetic instability and phenotype loss, due to the presence of repetitive DNA sequences. Engineering cellular phenotypes often requires the regulation of many genes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
